I am pleased to announce that we have recently published a comment paper in the Tasman Medical Journal in response to an editorial review by the journal's Editor-In-Chief concerning the use of hydroxychloroquine in the treatment of COVID-19. Despite expressing disagreement with the editorial, the Editor has been genuinely interested in publishing a robust academic debate. His handling of our manuscript was very professional and meticulous.
The study concisely reviews previously published work, so there are no new results presented. However, the importance of this study is that it provides a very concise statement of what should have been widely known and understood, by now, about pandemic response and specifically about the Raoult and Zelenko hydroxychloroquine-based multidrug protocols.
Some of the key points that we make are as follows:
1. Randomized controlled trials (RCTs) require large sample sizes to ensure sufficient randomization. RCTs with insufficient randomization cannot claim superiority over retrospective observational controlled studies.
2. There was sufficient evidence to justify the continuation of hydroxychloroquine-based multidrug treatments on an emergency basis by the end of April 2020, contrary to the claims of French regulators, currently preoccupied with persecuting Dr. Didier Raoult.
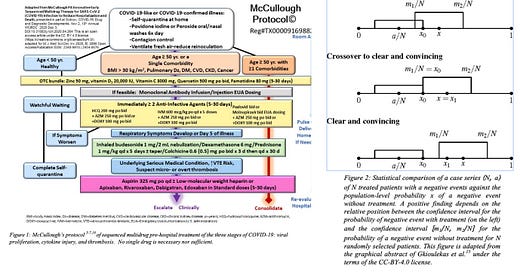
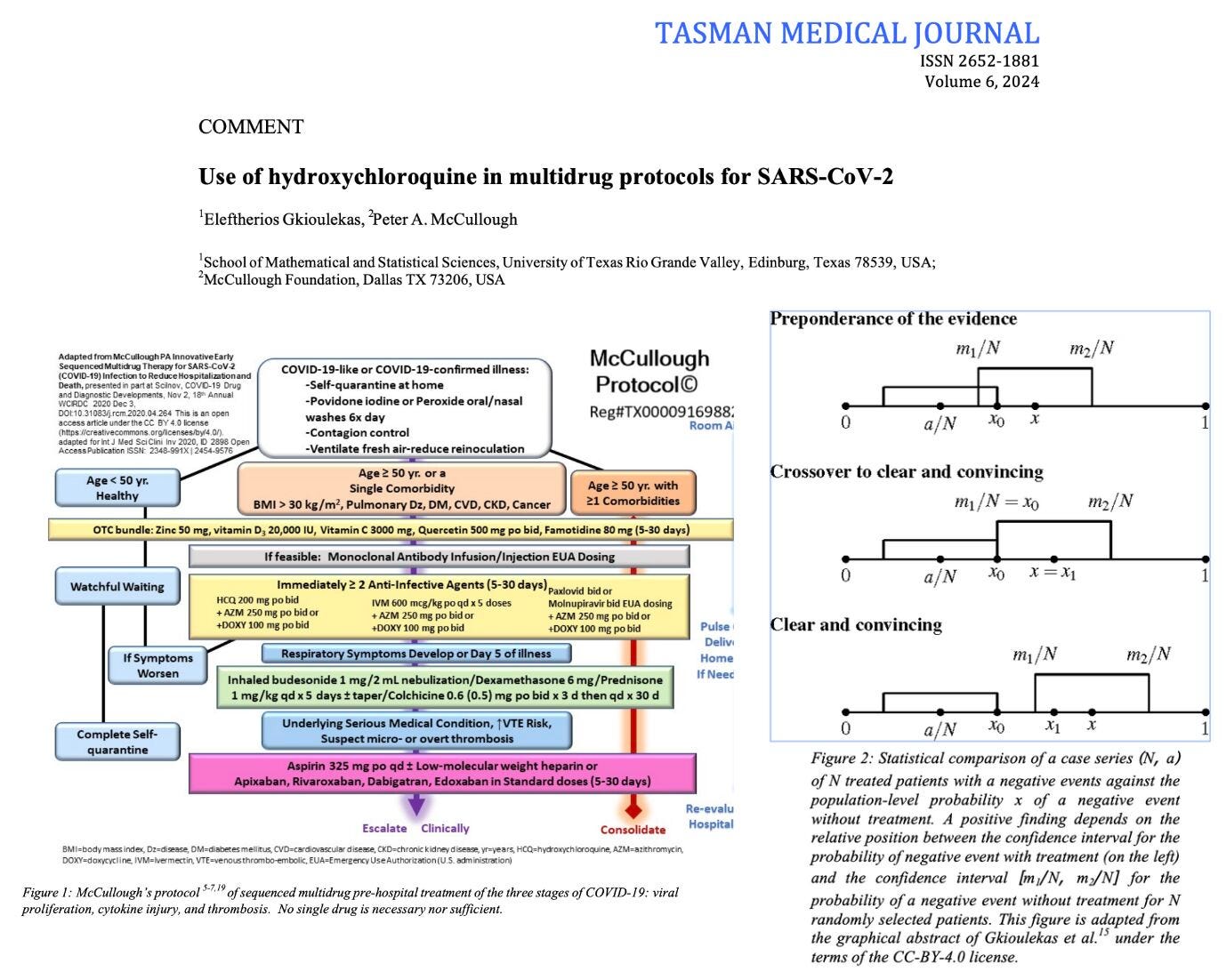
3. By December 2020, the evidence was sufficiently strong that there was no longer equipoise to ethically justify any randomized controlled trials against placebo. The crossover point for the failure of equipoise, based on Zelenko's data, was during June 2020.
We concluded the study with the following observations:
It is our interpretation that hydroxychloroquine played an important role in preventing hospitalizations and deaths due to COVID-19, particularly in 2020 with the more virulent strains. Widespread use of nasal sprays and gargles, aspirin, vitamin D, ivermectin, nirmatrelvir/ritonavir, molnupiravir, favipiravir, colchicine, corticosteroids, and anticoagulants in protocols all contributed to the benefits of early treatment which were widely favored over therapeutic nihilism in the pre-hospital phase. In case of a future pandemic, involving a novel disease, doctors should be encouraged to attempt treatments with repurposed medications based on biological plausibility, signals of benefit, and acceptable safety. Article 37 of the 2013 Helsinki declaration allows the use of unproven treatments if "proven interventions do not exist or other known interventions have been ineffective" and the unproven treatment "offers hope of saving life, reestablishing health or alleviating suffering". When these efforts result in case series of treated patients that show a large magnitude of benefit, then statistical comparison with historical controls can be used to support the strength of association between treatment and improved outcomes. As evidence accumulates, the Bradford Hill criteria framework can be used to assess the support for a causality claim, as an inference to the best explanation. This evidence can be gathered rapidly and form the basis for an agile emergency response to future pandemics, if public health is willing to leverage the clinical experience of medical doctors at the front lines.
I am grateful to Dr. Peter McCullough for joining with me in this study, and for his invaluable help and mentoring over the past several years. Special thanks to the late Dr. Zev Zelenko and the IHUM group (Dr. Didier Raoult, Dr. Phillip Brouqui, Dr. Mathieu Million, and colleagues) for pioneering the early hydroxychloroquine-based multidrug protocols. Furthermore, special thanks to Dr. Harvey Risch for pioneering the first epidemiological analysis of these protocols, and to Dr. Christian Perrone's research group for leading the independent reanalysis of Raoult's two-year dataset.
References
1. A. Millar, "The myth of hydroxychloroquine in the treatment of SARS-CoV-2 infection", Tasman Medical Journal 6(3) (2024), 13-16
2. E. Gkioulekas, P.A. McCullough: "Use of hydroxychloroquine in multidrug protocols for SARS-CoV-2", Tasman Medical Journal 6(4) (2024), 27-32
3. E. Gkioulekas, P.A. McCullough, V. Zelenko: "Statistical analysis methods applied to early outpatient COVID-19 treatment case series data", COVID 2(8) (2022), 1139-1182
4. M. Million, J-C. Lagier, H. Tissot-DuPont, I. Ravaux, C. Dhiver, C. Tomei, N Cassir, L. DeLorme, S. Cortaredona, S. Gentile, E. Jouve, A. Giraud-Gatineau, H. Chaudet, L. Camoin-Jau, P. Colson, P. Gautret, P-E. Fournier, B. Maille, J-C. Deharo, P. Habert, J-Y. Gaubert, A. Jacquier, S. Honore, K. Guillon-Lorvellec, Y. Obadia, P. Parola, P. Brouqui, D. Raoult, IHU COVID-19 Task Force. "Early Treatment with Hydroxychloroquine and Azithromycin in 10,429 COVID-19 Outpatients: A Monocentric Retrospective Cohort Study", Reviews in Cardiovascular Medicine 22 (2021), 1063-1072
5. P. Brouqui, M. Million, P. Parola, P.A. McCullough, D. Raoult. "Outcomes after early treatment with hydroxychloroquine and azithromycin: An analysis of a database of 30,423 COVID-19 patients", New Microbes and New Infections 55 (2023), 101188
6. V. Lounnas, E. Gkioulekas, M. Rendell, A. Lacout, X. Azalbert, C. Perronne, "An independent analysis of a retrospective cohort of 30,423 Covid-19 patients treated at IHU-Mediterranean in Marseille, France: Part 1, Efficacy of early treatment with hydroxychloroquine and azithromycin", Archives of Microbiology and Immunology 8 (2024), 51-66
7. H.A. Risch. "Early Outpatient Treatment of Symptomatic, High-Risk Covid-19 Patients that Should be Ramped-Up Immediately as Key to the Pandemic Crisis", American Journal of Epidemiology 189 (2020), 1218-1226
8. H.A. Risch, "Plausibility, not science, has dominated public discussions of the COVID pandemic", American Journal of Economics And Sociology 82 (2023), 411-424



I use the Zelenko protocol to this day, far superior to the vax, without the deadly side effects. Speaking of the vax, more people I personally know have died from the adverse effects and long term damage(heart damage, cancer and aggressive recurrent cancer , strokes and sudden death than ever died from covid. Not to mention the increased incidence of serious heart arrhythmias, tinnitus, autoimmune disorders etc. the vax is a graphic example of the cure being worse the disease. By orders of magnitude. All these deaths and damage from the vax are not only being ignored but actually systematically obscured by avoiding the study of readily available data and actually blocking access to relevant data. I believe that in the future, it will be proven that the Covid vaccine has been a medical holocaust
Published in 2005 before the medical institutions, journals, associations, and weak emasculated greedy compliant physicians started lying about HCQ:
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1232869/